International Journal of Veterinary Science and Research
Clinical safety of amino acids and vitamins administered through parenteral route
1Department of Preventive Veterinary Medicine, College of Veterinary Medicine, São Paulo State University, Jaboticabal, SP, Brazil
2Laboratory of Pharmacology and Clinical Pathology, College of Veterinary Medicine, Camilo Castelo Branco University, Descalvado, SP, Brazil
Author and article information
Cite this as
de Moraes AC, Scherma MR, da Rosa Prado EJ, de Faria VP, da Silva IC, et al. (2017) Clinical safety of amino acids and vitamins administered through parenteral route. Int J Vet Sci Res. 2017; 3(2): 55-61. Available from: 10.17352/ijvsr.000022
Copyright License
© 2017 de Moraes AC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.The parenteral use of amino acids and vitamins has been used to improve performance in animal production. However, few studies demonstrating the toxicity of these compounds. The present investigation evaluated the toxicological and clinical safety of the injectable supplement compound by amino acids and vitamins, administered by intramuscular route in Wistar rats. 56 females (± 220g), pubescent virgins, were randomly divided into seven groups (n=8): Treated with dose of 0.2, 2 and 4 mL,sampled 24 and 96 hours post-treatment (HPT), totaling six groups and one control group (Physiology Standard). Signifi cant increase in the enzymatic activity of serum ALT, AST, LDH and ALP was observed in animals treated with 2 and 4 mL (24 HPT). However, these changes returned to baseline levels 96 HPT, except in serum AST and ALP in animals treated with 4 mL. CK-NAC showed dose-response effect,since the higher compound dose was followed by proportional increase in the circulating levels of this enzyme, associated to muscle degeneration and necrosis area in rats treat with 4mL at 96 HPT. The evaluation of hepatic function did not result in signifi cant changes in the serum levels of triglycerides,cholesterol, albumin, urea and creatinine in the comparison between rats treated with 0.2 mL and control rats, demonstrating the innocuousness of this treatment on the activity of liver and kidney. However,deleterious effects were observed in animals subjected to doses of 2 and 4 mL, including observed increased splenic and hepatosomatic response, signifi cant decrease in circulating albumin levels and signifi cant leukocytosis, which is characterized by neutrophilia, followed by lymphopenia demonstrating the compound irritating effect on muscle tissue.
The amount of essential amino acids available in the metabolism must be iqual nutritional needs, seeking to supply maintenance and production requirements [1]. To improve the metabolic activity and productive performance of animals, the use of parenteral administration of amino acids and vitamins in animal production has increased mainly in ruminants (bovines, goats and sheep), due to the peculiarities of intermediary metabolism, resulting from biotransformations that foods suffer through ruminal fermentation [2].
A relevant aspect in the study of exogenous administration of organic modifiers should be related to the compounds safety. Therefore, these formulations need to provide clinical safety for animals. According to Castro et al. [3], all substance is potentially toxic, the correct dose differentiates a remedy of a toxicant. The practice of amino acids parenteral administration presents toxicological risks as observed with most chemical compounds such as spoiled food, industrial residues, among others, which induce lipid peroxidation mediated by the presence of free radicals, resulting in damage to biomembranes, consequently, promoting cellular and tissue dysfunctions [4,5].
Peroxidative processes involving lipids present in cell membranes represent critical factors in the pathogenesis of diseases related to free radicals, including inflammation, hepatic and renal disorders [6,7]. In the pathophysiology of these events, hepatocytes play a central role in the homeostatic balance of essential biological processes, regulating important metabolic functions involving the synthesis, activation, storage and catabolism of endogenous and exogenous chemicals [8]. According to Rivera-Huizar et al. [9] and Belo et al. [5], the occurrence of development of hepato-renal syndrome is high in cases of poisoning. Therefore severe damages to liver by different etiologies can impair the renal function and aggravate the clinical status of the animal.
Moreover, evidences indicate that the processes of hepatic regeneration depend on the availability of amino acids, vitamins, among others. Studies have shown the involvement of beneficial effect of leucine, isoleucine and valine supplementation for improving glucose metabolism in rats with cirrhosis [10, 11], as well as the synergistic effect of these nutrients, Kröger et al. [12] investigated the beneficial participation of methionine supplementation when combined with nicotinamide in reducing toxic effects induced by the treatment of methotrexate.
The growing use of parenteral supplementation with amino acids and vitamins in intensive production systems, seeking to improve the animals metabolic activity and productive performance, associated with the scarce literature regarding the possible side effects of this practice, this study aimed to evaluate the toxicological and clinical safety of amino acids and vitamins administered by intramuscular route in rats.
Materials and Methods
Animals
The experiment was conducted in the Laboratory of Clinical Pathology and Animal Pharmacology at the Veterinary Hospital, University Camilo Castelo Branco - UNICASTELO, approved by the Institutional Ethics Committee for Researches (process number 3042-3262/10), according to experimental protocols for good clinical practice [13]. 56 females albino Wistar rats (± 220g), pubescent virgins, born in the same period and from UNESP-Botucatu biotherium, were kept in polypropylene boxes, suitable for the species, fed with commercial feed (Labina® - Purina ) and water ad libitum, exposed to temperature and photoperiod of about 14 hours.
Experimental design
Animals were randomly divided into seven groups (n=8): Treated with dose of 0.2, 2 and 4 mL, sampled 24 and 96 hours post-treatment (HPT), totaling six groups and one control group (Physiology Standard). The treatments with the supplement of amino acids and vitamins (Table 1) were performed by intramuscular administration in the right hind limb. Likewise, the physiological standard group was submitted to the administration of saline 0.9% of 02.mL, to standardize the stress stimulus for capture and compound application.
Anesthesia and blood collection
The animals were anesthetized with an association of ketamine 10% (Vetaset® - Fort Dodge) and xylazine 2% (Rompun® - Bayer) in the proportion of 6:1, administered by intraperitoneal route at a dose of 1mL/kg of b.w., to collect blood samples from the posterior vena cava. Blood collection was realized in two sets of 5 mL syringes (needles 25x8), the first set was heparinized to collect plasma and second without anticoagulant to obtain serum.
Serum biochemistry
After centrifuging the blood samples without anticoagulant (5000 rpm for 10 minutes), the serum was separated for the determination of aspartate aminotrasnferase (AST), alanine aminotransferase (ALT), gamma glutamyl transferase (GGT), alkaline phosphatase (ALP), Lactate Dehydrogenase (LDH), albumin, urea, creatinine, total and direct bilirubin, creatine kinase (CK-Nac), creatine kinase MB (CK-MB), Triglycerides and Cholesterol, performed in enzymatic and colorimetric semi-automatic biochemical analyzer (Model LabQuest® - Bioplus Company).
Hematology
For determination of erythrogram was used automatic blood cell counter (Model CC510, Celm). The Leukocyte and platelet counts were performed manually in Neubauer chamber. The percentage of cell volume was determined in micro-hematocrit capillary tubes, centrifuged 5 minutes at 3000 rpm. Blood extensions were prepared for differential leukocyte count, stained with May-Grünwald-Giemsa-Wright. 200 cells were counted by extension to determine the percentage of each cell type [14].
Morphometric evaluation
At necropsy, liver, kidney and spleen were colleted for morphometric evaluation which was carried out according to Weibel et al. [15], using the ratio between the organ weight (OW) and the body weight (BW). These were expressed as liver somatic index (LSI), kidney somatic index (KSI) and spleen somatic index (SSI), and were calculated using the formula: Somatic Index (SI) = OW x 100 / BW.
Histological examinations
For histopathologic assessment, samples of liver, kidney, spleen and skeletal striated muscle from the right hind limb (administration site of the medicinal compound) were collected from all animals and fixed in 10% buffered formalin. After routine processing, the tissue was embedded in paraffin, sectioned at 5µm, and stained with hematoxylin and eosin (H&E) for photomicroscopic assessment. A blinded experienced pathologist performed histopathologic analyses and the histological findings related to tissues injury.
Statistical analysis
All data was statistically analyzed using a completely randomized split-plot design, with the main treatments (four doses 0, 0.2, 2 and 4 mL) and the subplots (two harvest times). Comparison of the different experimental groups was carried out by applying a PROC MIXED procedure, using the Statistical Analyses System [16]. Significant differences (P< 0.05) were estimated on the basis of Tukey´s test, according to Snedecor and Cochran [17].
Result
In the biochemical serum (Table 2), it was observed a significant increase (P <0.05) in the enzymatic activity of serum ALT, AST, LDH and ALP, mainly in animals treated with 2 and 4 mL, 24 hours after inoculation of the compound. However, these changes returned to baseline levels after 96 hours, except in serum AST and ALP in animals treated with 4 mL which remained statistically elevated compared to the control group. The group treated with 0.2 mL showed only a significant increase in the activity of ALP when compared to the control group. There was no significant variation (P> 0.05) on serum-enzymatic activity of GGT among different treatments.
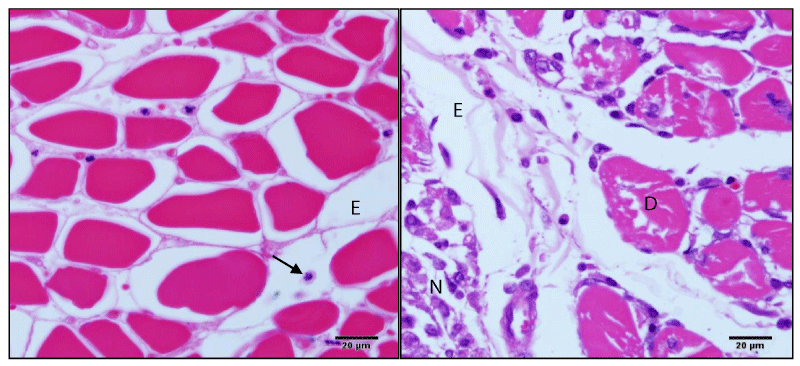
The study of serum CK-NAC showed dose-response effect, since the higher compound dose was followed by proportional increase in the circulating levels of this enzyme, corroborating this finding the Figure 1 presents the muscle degeneration and necrosis area in rats treat with 4mL at 96 hours after compound inoculation. Therefore the analysis of CK-MB did not present significant (p>0,05) changes in rats submitted to different treatments (Table 2). It was not observed clearly the effect of different treatments on serum levels of total and direct bilirubin (Table 2). However, a significant decrease of serum total bilirubin levels was observed in animals treated with 4 mL, 24 and 96 hours after compound inoculation, in comparison with the control group.
In the study of liver function, there was significant decrease in serum albumin levels 96 HPT in animals treated with doses of 2 and 4 mL, being accompanied by a decrease in triglyceride levels, although these results are not significant (P> 0.05). On the other hand, there was a significant increase in blood levels of glucose and cholesterol in animals treated with 2 and 4 mL (24HPT). Other relevant data reported in table 2 refer to the increase in blood levels of urea after 96 HPT in rats subjected to a dose of 4 mL, and no significant changes were found in the study of creatinine.
In the study of the red blood cells (Table 3), there was no significant change (P> 0.05) in the number of erythrocytes, percentage values of hematocrit, mean corpuscular volume (MCV), hemoglobin concentration and mean corpuscular hemoglobin (MCHC) when comparing animals treated with the dose of 0.2 mL and control rats. However, there was a significant decrease in the erythrocyte global counts, hematocrit values and the amount of hemoglobin concentration in rats subjected to a dose of 4 mL.
There was a significant thrombocytopenia in animals treated with 2 and 4 mL (24 HPT). With clinical evolution (96 HPT), these animals showed a significant increase in the number of circulating platelets (Table 4). The study of white blood cells showed increase (P <0.05) in the number of circulating leukocytes in all treated animals treated when compared to control rats in the acute response 24 HPT (Table 4). In the differential leukocyte counts, there was an increase (p <0.05) in the number of neutrophils in both relative and absolute values (Table 4). Figure 1 demonstrates accentuated inflammatory infiltrate, predominantly polymorphonuclear cells, and edema. The neutrophilia was accompanied by relative lymphopenia. No significant changes were observed in the monocyte, basophil and eosinophil counts (Table 4).
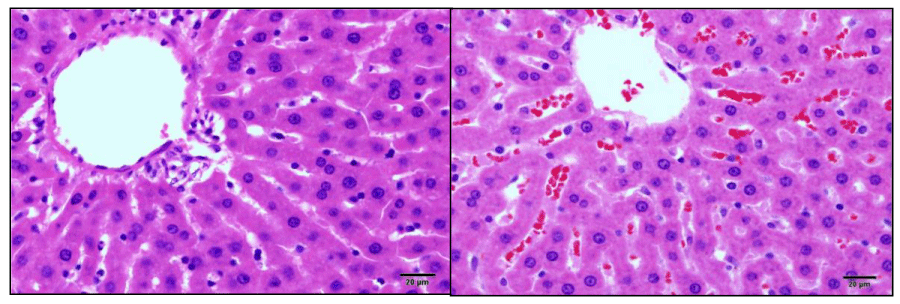
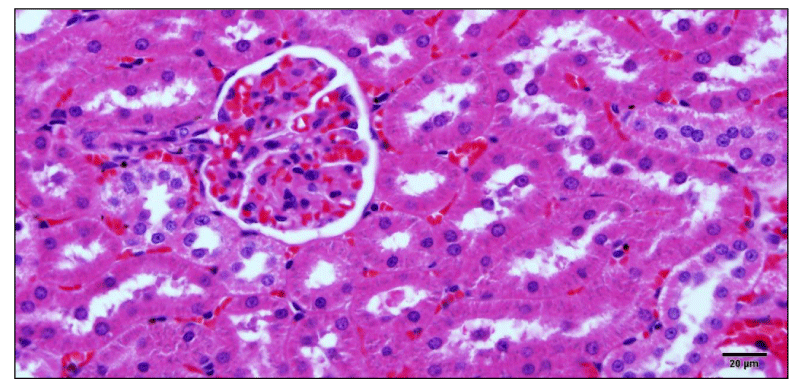
The hepatosomatic index evaluation showed a significant increase (P <0.05) in the weight of treated animals’ livers, this effect being much more significant in the animals treated with doses of 2 and 4 mL (HPT 24). Only animals treated with 4 mL showed an increase in liver size 96 HPT, these changes were accompanied by a significant increase (P <0.05) in the spleen somatic index (Table 5). Figure 2 and 3 show congestive changes in rats treated with 4mL (96 HPT) observed in liver and kidney, respectively.
Discussion
Toxic effects were mainly observed in animals treated with 2 and 4 mL. Increase in the enzymatic activity of serum ALT, AST, LDH and ALP was observed in animals treated with 2 and 4 mL. In biochemical assessment of liver activity, the serum enzymatic activity of ALT, AST, LDH, ALP and GGT can reveal hepatocellular injury and cholestatic changes [18,19]. According to Wills and Asha [20], increased serum enzyme activity of ALT and AST result of reversible or irreversible changes in the permeability of the hepatocyte cell membrane, the microsomal enzyme induction or structural lesion of the hepatobiliary ischemia, necrosis or cholestasis.
The serum CK-Nac study demonstrated a dose response effect, since animals submitted to higher doses were higher the circulating levels of this enzyme. On the other hand, the results of CK-MB analysis discards the possibility of changes in the myocardium, suggesting that elevated levels of CK-Nac is really resulted by the striated skeletal muscle lesion. The histopathological study in the application site corroborates these findings, because it was observed an intense inflammatory reaction, necrosis and muscle degeneration.
The hepatic functional tests can be determined by the study of substances synthesized in the liver or belonging to metabolic processes, such as albumin, BUN, glucose, cholesterol, triglycerides, among other [18,19,21]. In liver functional assessment, there were no significant changes in serum triglycerides, cholesterol, albumin and creatinine in the comparison between rats treated with 0.2 mL and control animals, occurring only transitory reduction in circulating values of urea and total bilirubin. According to Center [22], changes in energy metabolism are usually present in cases of acute liver failure. Moreover, the high concentration of glucocorticoids due to handling stress and treatment could favor energy metabolism by stimulating gluconeogenesis, increasing the bioavailability of free fatty acids resulting from the breakdown of triglycerides [22,23]. Sakabe et al. [24], Belo et al. [25,26] and Castro et al. [27] reported the occurrence of stress in animals, resulting of handling and treatment. According to these authors, the organic response to stress conditions is complex, involving neuro-endocrine mechanisms with the release of glucocorticoids and catecholamines which act by increasing the metabolic activity and consequently altering the organic balance.
The liver is essential for the homeostasis of proteins, and the primary site for synthesis of the majority of plasma proteins. Albumin is the main protein of hepatic export and represents about 50-60% of plasma proteins and about 75% of the oncotic pressure [19]. In this study, there were no changes in the circulating albumin values in rats treated with a dose of 0.2 mL, suggesting the hypothesis that treatment with the organic moderator did not impair the metabolic activity of the protein hepatic synthesis. However, deleterious effects were observed in animals submitted to doses of 2 and 4 mL which showed a significant decrease in circulating albumin levels.
On the other hand, 90% of ammonia released in the liver is converted into urea which becomes the main metabolic product in liver detoxification protein catabolism [9]. The study of blood urea and creatinine levels showed normal values when comparing animals treated with saline and 0.2 mL. Despite the kidney somatic study did not vary significantly, congestive changes and tubular cell swelling were observed in renal histopathology of animals treated with 4 mL.
The erythrocyte and platelet study of rats treated with 0.2 mL showed no significant changes compared with control animals throughout the experimental period, with the mean values observed within the normal range for Wistar rats described by Fukuda et al. [28]. However, there was significant decrease in the global number of erythrocytes, hematocrit values and in the amount of circulating hemoglobin in rats treated with 4 ml, characterizing a normocytic / normochromic anemia, since there were no alterations in the values of MCV and MCHC.
However, there was a significant leukocytosis in animals treated with the amino acid compound, which is characterized by neutrophil, followed by lymphopenia, demonstrating the compound irritant action in the muscle tissue. In acute inflammatory reactions occurs significant increase in the number of neutrophils that are recruited by chemotaxis to act in the inflamed site [29,30].
Animals with liver lesions may present functional changes in the number and function of platelets. In cases of intra and extrahepatic circulatory deviations, patients may present with congestive splenomegaly, resulting from portal hypertension, leading to conditions of thrombocytopenia [19]. This correlation was evident in this study, since animals treated with a dose of 4 mL showed increased splenic and hepatic somatic index, as well as congestive changes in liver and kidney tissue.
During the experimental period, the amino acids compound treatment in different doses did not result in clinical signs and behavioral changes in rats that could suggest the occurrence of toxic effects, and the recommended dose (0.2 mL) demonstrated to be clinically safe.
The authors thank the Camilo Castelo Branco University and Laboratory Aminogel by fomenting this study.
- Broderick GA, Wallace RJ, Orskov ER (1991) Control of rate and extent of protein degradation. In: Tsuda T, Sasaki Y, Kawashima R (Ed.) Physiological aspects of digestion and metabolism in ruminants. New York: Academic Press 541-592. Link: https://goo.gl/Qjg8jM
- Alves D (2004) Nutrição Aminoacídica de Bovinos. Revista Brasileira de Agrociência. 10: 265-271. Link: https://goo.gl/bWVwrM
- Castro MP, Claudiano GS, Petrillo TR, Shimada MT, Belo MA, et al. (2014) Acute aerocystitis in nile tilapia bred in nete cages and supplemented with chromium carbochelate and Saccharomyces cerevisiae. Fish Shellfish Immunol 31: 284-290. Link: https://goo.gl/FMwUYa
- Cho EJ1, Yokozawa T, Rhyu DY, Kim HY, Shibahara N, et al. (2003) The inhibitory effects of 12 medicinal plants and their component compounds on lipid peroxidation. Am J Chin Med 31: 970. Link: https://goo.gl/LSRpWQ
- de Andrade Belo MA1, Soares VE, de Souza LM, da Rosa Sobreira MF, Cassol DM, et al. (2012) Hepatoprotective treatment attenuates oxidative damages induced by carbon tetrachloride in rats. Exp Toxicol Pathol 64: 155-165. Link: https://goo.gl/ZKpdDy
- Halliwel B (1997) Antioxidants and human disease: a general introduction. Nutr Rev 55: 44-52. Link: https://goo.gl/Xpa4ZL
- Datta S, Basu K, Sinha S, Bhattacharyya P (1998) Hepatoprotective effect of a protein isolated from Cajanus indicus (Spreng) on carbon tetrachloride induced hepatotoxicity. Indian Journal of Experimental Biology 36: 175-181. Link: https://goo.gl/JHgSzu
- Rao GM, Rao CV, Pushpangadan P, Shirwaikar A (2006) Hepatoprotective effects os rubiadin, a major constituent of Rubia cordifolia Linn. J Ethnopharmacol 103: 484-490. Link: https://goo.gl/9dDJCm
- Rivera-Huizar S, Rincón-Sánchez AR, Covarrubias-Pinedo A, Islas-Carbajal MC, Gabriel-Ortíz G, et al. (2006) Renal dysfunction as a consequence of acute liver damage by bile ligation in cirrhotic rats. Exp Toxicol Pathol 58: 185-195. Link: https://goo.gl/DVXfUz
- Habu D, Nishiguchi S, Nakatani S, Kawamura E, Lee C, et al. (2003) Effect of oral supplementation with branched-chain amino acid granules on serum albumin level in the early stage of cirrhosis: a randomized pilot trial. Hepatol Res 25: 213-318. Link: https://goo.gl/GhSRr5
- Nishitani S, Takehana K, Fujitani S, Sonaka I (2005) Branched-chain amino acids improbé glucose metabolism in rats with liver cirrosis. Am J Physiol Gastrointest Liver Physiol 288: 1292-1300. Link: https://goo.gl/f89eFT
- Kröger H1, Hauschild A, Ohde M, Bache K, Voigt WP, et al. (1999) Nicotinamide and methionine reduce the liver toxic effect of methtrexate. Gen Pharmacol 33: 203-206. Link: https://goo.gl/CNegWn
- EMEA- European Medicines Agency (2000) Guideline de Boas práticas Clínicas. The European Agency for the evaluation of medicinal products. Link: https://goo.gl/BEkCyg
- Belo MAA, Souza DGF, Faria P, Moraes FR, Onaka EM (2013) Haematological response of curimbas Prochilodus lineatus, naturally infected with Neoechinorynchus curemai. Journal of Fish Biology 82: 1403-1410. Link: https://goo.gl/jUUVVz
- Weibel ER, Stäubli W, Gnägi HR, Hess FA (1969) Correlated morphometric and biochemical studies on the liver cell. J Cell Biol 42: 68-91. Link: https://goo.gl/uyme61
- SAS Institute Inc (2001) SAS/STAT software changes and enhancements though [computer program]. Release 8.2. Cary: SAS Institute. Link: https://goo.gl/55fKt7
- Snedecor GW, Cochran WG (1980) Statistical Methods. Iowa State University Press, Iwoa, USA: 75.
- Suja SR, Latha PG, Pushpangadan P, Rajasekharan S (2004) Evaluation of hepatoprotective effects of Helminthostachys zeylanica (L.) Hook against carbon tetrachloride-induced liver damage in Wistar rats. J Ethnopharmacol 92: 61-66. Link: https://goo.gl/BV1PT3
- Shih CC, Wu YW, Lin WC (2005) Aqueous extract of Anoectochilus formosanus attenuate hepatic fibrosis induced by carbon tetrachloride in rats. Phytomedicine 12: 453-460. Link: https://goo.gl/ACNMgR
- Wills PJ, Asha VV (2006) Protective effect of Lygodium flexuosum (L.) Sw. extract against carbon tetrachloride-induced acute liver injury in rats. Journal of Ethnopharmacology 108: 320-326. Link: https://goo.gl/aPt5zn
- Kerr MG (2003) Bilirrubina e metabolismo Lipídico. In: KERR MG Exames Laboratoriais em Medicina Veterinária: Bioquímica Clínica e Hematologia. 2ª Ed., São Paulo, Roca 149-158. Link: https://goo.gl/tSRbps
- Center SA (1992) Fisiopatologia e Diagnóstico laboratorial das moléstias hepáticas, In: Tratado de Medicina Interna Veterinária. 3. ed. São Paulo: MANOLE 9: 1487-1546.
- Belo MA1, Schalch SH, Moraes FR, Soares VE, Otoboni AM,et al. (2005) Effect of dietary supplementation with vitamin E and stocking density on macrophage recruitment and giant cell formation in the teleost fish, Piaractus mesopotamicus. J Comp Pathol 133: 146-154. Link: https://goo.gl/S5Z1RE
- Sakabe R, Moraes FR, Belo MAA, Moraes JER, Pilarski F (2013) Kinects of chronic inflammation in Nile tilapia supplemented with essential fatty acids n-3 and n-6. Pesquisa Agropecuária Brasileira 48: 313-319. Link: https://goo.gl/ggiMdV
- Belo MAA, Moraes JRE, Soares E, Maritns ML, Brum CD, et al. (2012) Vitamin C and endogenous cortisol in foreign-body inflammatory response in pacus. Pesquisa Agropecuária Brasileira (1977. Impressa) 47: 1015-1021. Link: https://goo.gl/gbk7Zs
- Belo MAA, Moraes FR, Yoshida L, Prado EJR, Moraes JRE, et al. (2014) Deleterious effects of low level of vitamin E and high stocking density on the hematology response of pacus, during chronic inflammatory reaction. Aquaculture 422-423: 124-128. Link: https://goo.gl/HQqH9U
- Castro MP, Claudiano GS, Bortoluzzi NL, Garrido E, Fujimoto RY, et al. (2014) Chromium carbochelate dietary supplementation favored the glucocorticoid response during acute inflammation of Piaractus mesopotamicus. Aquaculture 432: 114-118. Link: https://goo.gl/9tYxM8
- Fukuda S, Tsuchikura S, Iida H (2004) Age-related changes in blood pressure, hematological values, concentrations of serum biochemical constituints and weights of organs in the SHR/Izm, SHRSP/Izm and WKY/Izm. Experimental animal 53: 67-72. Link: https://goo.gl/jQ5D3M
- Belo MAA, Souza LM, Soares E, Sobreira MFR , Cassol DMS, et al. (2009) Tratamento hepatoprotetor favorece a resposta leucocitária de ratos Wistar intoxicados por CCL4. Archives of Veterinary Science 14: 74-82. Link: https://goo.gl/XfmTLC
- Reque R, Moraes JRE, Belo MAA, Moraes FR (2010) Inflammation induced by inactivated Aeromonas hydrophila in Nile tilapia fed diets supplemented with Saccharomyces cerevisiae. Aquaculture 300: 37-42. Link: https://goo.gl/HBexxp
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
