International Journal of Veterinary Science and Research
Phenotypic Characterization and Predicting Body Weight of Local Chicken in Doba and Mesala Districts, West Hararghe Zone, Ethiopia
Hramaya University, School of Animal and Range Sciences, Ethiopia
Author and article information
Cite this as
Musa SA, et al. Phenotypic Characterization and Predicting Body Weight of Local Chicken in Doba and Mesala Districts, West Hararghe Zone, Ethiopia. Int J Vet Sci Res. 2026; 12(2): 8-16. Available from: 10.17352/ijvsr.000157
Copyright License
© 2026 Musa SA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.The study was carried out to describe the phenotypic characterization, and body weight prediction from LBM of local chicken ecotypes in the Doba and Mesala districts of the West Hararghe Zone, Oromia Regional State, Ethiopia. A total of 420 adult live chickens of both sexes, 210 chickens (92 male and 118 female) older than 24 weeks from Doba, and 210 chickens (65 male and 145 female) from Mesala were employed for gathering information on both quantitative and qualitative traits. Univariate analysis, multivariate analysis, Generalized Linear Model (GLM) procedure, Multiple Linear Regression (MLR), and Pearson Correlation Coefficient (PCC) of SAS (version 9.4) were used to analyze the quantitative data, while qualitative data of the investigated chicken were analyzed using the frequency procedure of the Chi-square (χ2) test and descriptive statistics. There were significant differences (p < .0001) in the diversity of colour between and within the agroecology concerning body plumage colour. However, in the Doba and Mesala districts, respectively, brown, white, and red comprise the majority of the local chicken ecotype colour in both females and males. The findings showed that morphological appearances (Head shape and comb color) varied significantly (p < 0.05. More desirable traits and community-based genetic improvement programs should be developed and implemented with the inclusion of phenotypic characterization and body weight improvement of local chicken breeds. These steps would complement the current study by conserving as well as using diverse indigenous chicken genetic resources sustainably.
The majority of the indigenous animal genetic resources from Asia to Africa are thought to enter Africa through Ethiopia, which is home to a sizable number of livestock populations around the globe [1]. According to EBI (2016), Ethiopia possesses seven local chicken ecotypes: the Farta, Horro, Jarso, Konso, Mandura, Tepi, and Tillili. The small number of breeds included in the database and several phenotypic, morphologic and genetic characterizations showed variations among ecotypes, and that most of the variations in the local chickens are attributed to variations within individual populations.
Identifying morphological characteristics of distinct populations and describing their production within a given production environment, including the description of breeds’ production environments and recording of their geographical distributions are generally referred to as phenotypic characterization of AnGR [2]. The local chicken of Ethiopia was named by their plumage color or geographic origin of sampling, as in many other parts of Africa. In Ethiopia, local chickens are found in huge numbers distributed across different agro ecological zones under a traditional family-based scavenging management system, and variations in morphological and morphometric traits are common among local chicken populations. However, the local chickens that have mainly been selected naturally or by the farmers who keep them for their adaptive fitness to a specific area are often poor in their egg production and characterized by late maturation as well as long broodiness due to the prevalence of diseases and predators, low genetic potentials, feed shortage and limited feed resources, constraints related to institutional, infrastructural, socio-economic and the economic contribution of local chicken is not proportional to their huge number [3].
Chicken genetic resource characterization is launched to measure their performance and to improve the production system in different parts of the country, as expressed in many livestock-related sectors. However, the lack of information on farmers’ breeding practices based on agro ecologies and the identification of trait preference for the breeding objective is creating difficulty in implementing and designing the chicken breeding program. Therefore, identifying adapted local chicken genotypes for market requirements, genetic improvement, and production circumstances through investigation should be conducted; many researchers like Andualem, et al. [4], Tadele, et al. [5], and Mustefa, et al. [6] have made phenotypic characterization of local chicken in some parts of the country. However, no previous studies have been carried out for the phenotypic and predicted body weight from LBMs in the Doba and Mesala districts. Thus, the present study sought first to characterize and predict body weight from LBMs in the study area. Therefore, this research was developed to address the following objectives.
- To describe the phenotypic characterization of local chicken, and
- To predict body weight from linear body measurements
Materials and methods
Data Collection procedure
Measurement of quantitative and qualitative traits of village chicken ecotypes was done and recorded, using a structured format for morphological description, following standard descriptors [7]. The minimum number of mature chickens required for phenotypic characterization is 10-30 cocks and 100-300 hens [7]. Based on this concept, from direct observation of sexually matured chickens and additional information from the owner, a total of 420 adult live chickens of both sexes: 210 chickens (92 male and 118 female) above 24 weeks of age from Doba and 210 chickens (65 male and 145female) from Mesala were used to collect quantitative and qualitative trait data such as plumage colour, comb type, comb size, presence or absence of spurs, shank colour, earlobe colour, eye colour, presence or absence of ear lobe, comb colour, skin colour, body shape, and head shape.
Quantitative traits like body weight (BW), body length (BL), chest circumference(CC), wing span (WS), shank length (ShL) and circumference (SC), keel length (KL), ear lobe length (ELL), spur length (SpL), beak length (BkL), comb length (CL) and height (CH) were measured using a textile measuring tape. A hanging spring balance was used to measure the live body weight of individual chickens from male and female adult live chickens based on the standard format breed descriptor list [7].
Sample size and sampling techniques
Before the actual survey work, a rapid field survey was made to locate the distribution of local chickens and their production system. The distribution and numbers of local chickens were then obtained from the Office of Agriculture and Natural Resources (OANR) of each district before starting the actual fieldwork. A multi-stage sampling procedure (purposive and random) was applied for the study; then, two districts were purposively selected because of chicken production potential and agroecology. A stratified sampling technique was employed to stratify Keeble’s into the two districts, the lowland and the highland. Three rural Keeble’s per district were selected purposively based on agroecology and chicken production potential.
A total of 200 respondents (102 from Doba and 98 from Mesala districts) were randomly selected from households included in the study questionnaire survey. Three focus group discussions per district, comprising six to ten members selected from the development agent of the Keeble and focal farmers who have good experience in chicken production from both sexes, were also conducted. The discussion was focused on a production system and breeding practices, marketing system, trait preferences, and major constraints of chicken production.
Sample size determination
The total number of households included in the study was determined according to the formula given by Arsham 2002. N = 0.25/SE2 Where, N = Sample size, SE = Standard error. Thus, using the standard error of 0.0353 with a 95% confidence level, 200 households were included in the study. The number of respondents (farmers) per single agroecology or Keeble was determined by proportionate sampling technique based on their household population size as follows:
W= [A/B] x No
Where: W = Number of respondents required per single agroecology
A = Total number of households (farmers) living per a single selected Keeble
B = Total sum of households living in all selected samples Keeble’s and
No = the total required calculated sample size (http://www.raosoft.com/samplesize.html).
Data management and statistical analysis
After data was collected, it was coded and recorded in a Microsoft excel sheet and made ready for analysis. Preliminary data analysis was employed before conducting the main data analysis for clearance and checked for any type of error that occurred during data collection which then was exported to SAS (version 9.4). Statistical analyses were made separately for male and female chickens on variables that varied in sex; otherwise, the data were merged and analyzed together.
Univariate analysis: The qualitative and quantitative data were analyzed by using SAS (version 9.4).ANOVA was also used to locate means that are significantly different. Discrete measurements of the qualitative traits of the investigated chicken were analyzed using the frequency procedure of the Chi-square (c2) test. Mean comparisons were made using Tukey’s studentized range test method at p < 0.05. Pearson correlation coefficient was used to estimate the correlation between linear body measurement and body weight. More specifically descriptive statistics and GLM was used for linear body measurements with the fixed effects of sex and agroecology. Sex and district class group of chicken was fitted as independent variables while body weight and other LBM were fitted as the dependent variable. A multiple linear regression procedure was also used to regress body weight for both sexes to determine the best-fitting regression equations for the prediction of live body weight. . Discrete measurements of the qualitative traits of the investigated chicken were analyzed using the frequency procedure of the Chi-square (c2) test. The following model was used for the measurement of data.
Model 1: Yij = μ + Di + Sj + DS ij +eij
Where: Yijk = the observed body weight and LBM of chickens in the ith district, jth sex, and ijth the interaction effects of the district by sex
μ = Overall mean
Di = Fixed effect of ith district (1= Doba and 2= Mesala)
Sj = Effect of jth sex (1= Male and 2= Female)
DSij = District and sex interaction effects
eijk = Random residual error
Model 2: Y = b0 + b1x1 + b2X2 +... b11X11
Where: Yj = Response variable or predicted mean body weight of chickens; b0 = the intercept; X1 + X11+ …X11are the explanatory variables for body length, chest circumference, keel length, shank circumference, wattle length, spur length, ear lobe length, beak length, shank length, comb length, and comb height, respectively. b1 + b2+ …b11 are the regression coefficient of the variables X1+ X2+ …X11
Results and discussions
Phenotypic characteristics of chickens
Qualitative traits: Plumage colours of various chicken ecotypes in the study areas are shown in Table 1 and Figure 1. From the result of correspondence analysis, brown plumage color, white, red, wheaten, and reddish-brown ecotype chickens are closer to Doba district while red colour chickens, reddish-brown, yellow-brown, and white ecotypes are closer to Masala district. Moreover, the present study showed that the major plumage color frequently observed in the Doba chicken ecotype is brown (34.75%), followed by white (16.10%) and wheaten (11.86%) in females, while white cocks (26.09%) followed by reds (18.48%) are predominant one. Whereas, major plumage colours for Masala chicken ecotype include red (25.71%), followed by reddish-brown (14.76%) and yellow-brown (9.52%). The reason that is more females and male with brown, white, and red plumage colors can be that farmers in the study area prefer to raise them. After all, there is a stronger market demand for them and consumers prefer them. This shows that farmers use some form of selection to exclude other plumage colours and that different genes control feather colors.
Similar findings have been reported about the brown, white, and red plumage colors, which seemed to be more frequent than other colors, in the Yabello and Dire districts of the Borena zone [8]. Amine, et al. [6] also described that higher frequency of red-colored chicken population in Southern Ethiopia, which is comparable with the current result found in Mesala District. However, Bogale, et al. [9] revealed inconsistent findings from the Darolabu district of the West Hararghe zone, where white with black plumage-colored chickens (13.2%) were found to be the most common. A mixture of white, black and red plumage colour was the predominant colour observed in GamoGofa zone [10]. Observed variations in the plumage colours are might be due to the results of genetic and environmental factors as well as artificial selections practiced by farmers in the study area.
In general, a considerable degree of colour variability was observed between and within agroecology; in the Doba and Masala districts, the majority of local chicken ecotype colours for both males and females were white and brown, and red respectively. Diversified plumage colours may represent a potential future for selection-based genetic improvement of local chicken ecotypes.
Morphological variation in local chicken ecotypes: BaBased on the identified dimensions of correspondence analysis the red eye color chickens from the Doba district were closely clustered together and had higher frequency (48.57%) while orange the orange eyes color is more closely to the Masala district and represented more frequency (82.38%) (Table 2). Brown eyes colour were equal dimensions in both ecotypes. This result is corroborated by the report of Ayana [11] who reported that the dominant eye color of the chicken ecotype was orange and red colour in the Awi zone Amhara regional state. Moreover, red eye colour is influenced by the degree of iridic vessel injection and the hemoglobin content of the blood [12]. According to Nelson [12], when chickens suffer from general anemia, a smaller amount of red coloration will be noted due to loss of blood volume. Therefore, eye colour variation depends on the pigmentation (carotenoid pigments and blood supply) of several structures within the eye [13]. Another variation might be nutritional status, a breed’s specific traits, genotype, and reflected adaptation fitness to their environment.
The plain and snakehead shapes were close to the Masala districts while the crest head shape was not close to both district. Overall, in both agro ecologies, 61.43% and 68.57% of plain head shapes were predominant followed by snakehead shapes in Doba and Masala ecotypes respectively. These findings are corresponding to the results reported by Getachew, et al. [14] in the Bench Maji zone. Contrasting results have also been reported by Shishay, 2014 who stated that the occurrence of crest-headed chickens (55.8%) was more frequent than plain-headed chickens. This might be due to the expression of the gene found in local chicken populations. The interpretation of correspondence analysis showed that the single comb type is closer to the Doba districts followed by the pea comb type while double followed by a single more approach to the Mesale districts. Single comb-type (62.85%) in Doba and double comb-type (34.28%) were found to be more represented in Masala. This result agreed with the result of Wario, et al. [8] carried out in Yabello and Dire districts of Borana zone. However, this contradicted the result of Andualem, et al. [4] who found 52.8% of chickens had rose comb while the rest 30.9% and 16.3% had pea and single respective comb-type in the Awi zone Amhara region. Variations between agro-ecologies might be associated with the traits preference and selection criteria of the farmers in the study area.
Regarding the comb color, red and pale comb colours were more closely to the districts in both respective agro ecologies. Small and medium comb sizes were more closely to the Doba district while small and large comb sizes approached the Masala Districts. Comb size is associated with gonadal development and intensity of light but comb type is the consequence of gene interaction (Bell, 2002). Generally, the results indicated that there were significant variations (p < 0.05) in morphological appearances (head shape and comb colour) while differences were significant at (p < .0001) in comb type, comb size, and eye color of local chicken in the study area. Association traits observed between ecotypes might be due to interbred population breeds for the past many years or transport of local chickens between agro-ecologies because of marketing at the border urban of Tullo district, which is a bridge between Mesala and Doba districts. And as well as random mating concerning traits, natural and artificial selection of chicken producers.
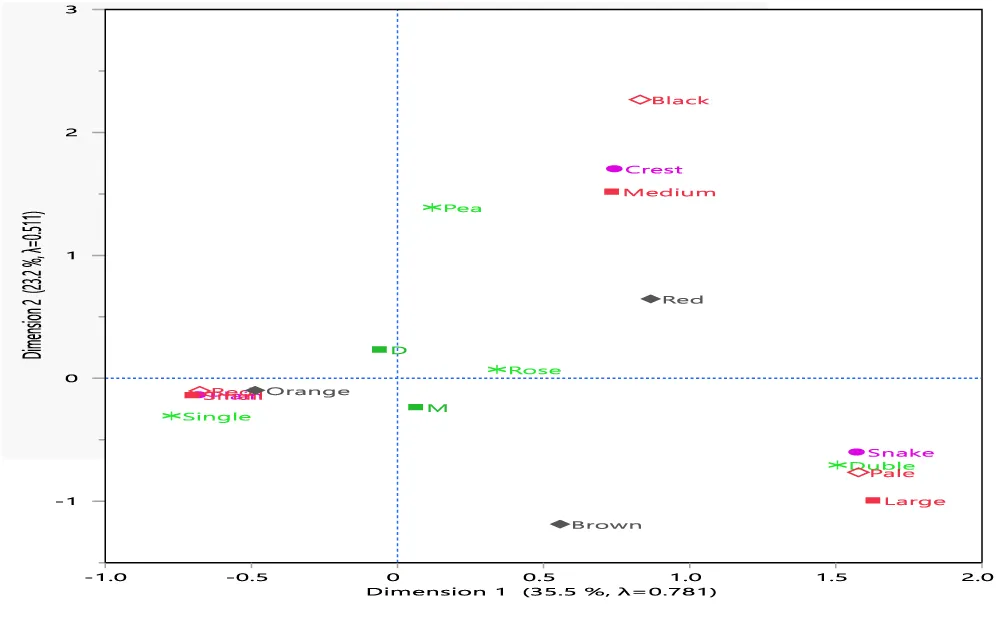
Multiple correspondence analyses: For the sample chicken population of the two districts, five of eleven qualitative morphological traits (i.e., head shape, comb type, comb colour, comb size, and eye colour) were found significant (p value < 0.05, Table 2). Multiple correspondence analysis of SAS (PROC CORRESP) was thus carried out on these five significant traits. To examine the association between the district and qualitative traits, the district was included in the analysis as a supplementary variable. Figure 2 shows a bi-dimensional graph representing the associations among the categories of the analyzed variables.
From Figure 2, it can be seen that 58.7% of the total variations are explained by the first two dimensions, 35.5% by the first, and 23.2% by the second dimensions. On the dimensions identified the sample chicken population of Mesela district clustered together with snake HS, double CT, pale CC, large CS, and brown EC; while the corresponding values of the Doba district do not show clustering characteristics like Mesela.
Live body weight and linear body measurements
Information on live body weight and linear body measurements of the existing breed types has a mandatory role in the selection programs. The body weight and linear body measurements for chicken ecotypes in the study districts are presented in Table 3.
Sex effect: The least-square means for the effect of sex was significant at (p < 0.05). Male chickens had significantly (p < 0.05) higher LSMs values in all studied traits while female indicates lower values than males in all traits. The average mature body weights of local male chickens (1.67 kg) were significantly higher (p < 0.05) than female chickens (1.34) Table 3. Also significantly (p < 0.05) higher mean values of body length (39.42 cm) were obtained from male chicken ecotypes. This result was in line with that of Wario, et al. [8], who reported that male local chickens had longer bodies than females (40.04) in the Yabello and Dire districts and also in line with the reports of Bekele, et al. [3] and Getu, et al. on different local chicken populations of Ethiopia. Such differences between males and females may be attributed to the differences in growth hormones.
District effect: The district had a significant effect (p < 0.05) on body weight and linear body measurements were the maximum value recorded from the Mesala chicken ecotype. Significantly (p < 0.05) higher least square mean values of body weight, body length, shank length, shank circumstance, ear lobe length, comb length, and wing span were recorded from Mesala chicken ecotypes. This result is different from the report of shisha 2014, who revealed that lower values for body weight and all other linear body measurements were obtained from highland chickens. The variation in body weight and other LBMs could be attributed to differences in the environment as well as management practices employed in the two districts or even it is due to the presence of strains or sub-populations within the breed.
Sex by district interaction: The interaction between sex and district was significant (p < 0.05) for chest circumstance, comb length, comb height, spur length, body length, body weight, and wing span. Significantly (p < 0.05) higher mean values of this parameter were obtained from the Masala district in both sexes of chicken ecotypes. This result is also in line with the findings of Emebet, et al. [15] who reported that higher LSMs values were obtained from male chicken ecotypes in three districts of South West and South parts of Ethiopia.
Generally, quantitative traits of local chicken in both agro ecologies showed significant variations in most of the studied traits. This variability of local chickens in the study area is a major indicator of the existence of high genetic variability among local chicken ecotypes which serves as a big potential for genetic improvements of the local chicken ecotypes through appropriate genetic improvement methods.
Diversity in measurable traits is an indication of their genetic variability in local chickens of the study area. This is due to different production environments in different agro ecological zones as a result of variations in the agro ecological variables.
Correlation of body weight and other LBMs of cocks and hens
The correlation coefficient between body weight and other LBMs for both male and female are shown in Table 4. Body weight of hens’ had a significant (p < 0.05) and mild positive correlation with wing span (0.31) and keel length (0.21) while the weak positive correlation with comb high (0.23), beak length (0.18), body length (0.20), chest circumstance (0.15) and shank length (0.17). But the correlation between body weight with comb length (0.08), spur length (0.07) and shank circumstance (0.10) is not significantly correlated. For cocks’ body weight had a significant (p < 0.05) and positive correlation with all traits except for wing span. Weak positive correlation between spur length (0.20) and keel length (0.15) while body length, chest circumstance, comb length, comb height, ear lobe length, shank length, and beak length were a mild positive correlation. This result was in line with the report of Tareke, et al. [16] who reported that body weight had a significantly positive correlation with body length, keel length, shank length, and chest circumference in the Bale zone. However, this result argues with the report of Ukwu, et al. [17] and Alabi, et al. 2012 that a strong positive correlation exists between body weight and linear body measurements in indigenous chickens of Nigeria and South Africa, respectively. The observed variation might be due to the pleiotropic effect and gene linkage effects between the two traits of indigenous chickens as well as environmental factors that are exposed to chickens in the study area.
In general, knowing a particular chicken breed’s linear body measures is crucial for economic evaluation in terms of its use and identification of the breed based on the other trait without additional expense and time. Overall, the relationship between LBMs and body weight allows us to estimate body weight and may be quickly applied to the selection and breeding of native chickens.
Prediction of body weight from linear body measurements
Body weight has been the pivot on which animal production thrives. The knowledge of livestock weight assessment remains the backbone on which all animal production management practices are hinged. There is often a great need for livestock herdsmen to know how much their animals weigh. Reasonable skill in estimating weight is, therefore, necessary for the stockman as it will frequently be necessary to know weights when the weighing scale is not readily available or its use is not practically feasible [18].
In this study to develop the prediction equation and predict body weight from LBMs, a multiple linear regression procedure was carried out separately for each sex based on eleven quantitative traits (BL, CC, ShL, CL, CH, EL, SpL, WL, ShC, KL, and BkL) which had a positive correlation with body weight. Variables that best fitted the model were selected using the selection criteria R2 (adj), RMSE, Cp statistics, Akaike’s Information Criteria (AIC), and Schwarz Bayesian Criteria (SBC). The best-fitted variables were selected using a higher value of R2 (adj), and a smaller value of Cp, AIC, SBC, and RMSE. The results of the MLR equations that utilized all possible regression models are given in Tables 5,6.
For the male chicken population, as shown in Table 5, Model 5 was chosen as it has the optimum combination of adjusted R2 and a smaller value of Cp, AIC, SBC, and RMSE. Whereas, for the female chicken population, Model 4 was chosen as it has the optimum combination of adjusted R2 and a smaller value of Cp, AIC, SBC, and RMSE (Table 5). In both cases especially the selection criterion Cp was efficient in selecting the optimum model. Thus, the prediction equation of body weight from LBMs could be done based on the following models:
y = 1.67 + 0.06CC + 0.05CL – 0.02SpL + 0.03WL – 0.02KL for male sample population and
y = 1.35 + 0.01BL + 0.02CH + 0.01EL + 0.01KL for female sample population respectively.
The best model is one that has the lowest C (p) while the coefficient of determination R2 (adj) shows the percentage of the total variability explained by the model, the small C (p) suggests accuracy and small variance in estimating the populations of the regression coefficient. Mean Square Error (MSE) decreases significantly as additional variables are added to the model; nevertheless, adding additional variables to the model can increase MSE. The values of the coefficients of determination R2 (adj) ranged from 0.22 to 0.36, with CC +CL+ SpL+ WL+ KL showing the highest R2 (adj) value for males. This combination of parameters is significant (p < 0.05) for male chickens and the best predictor of body weight in the study area. The small C (p) value and highest R2 (adj) value for female chickens were explained by BL +CH +EL +KL equation. This result is different from the observations reported in Nigerian indigenous chickens [17] and Fikrineh [19] in Ethiopia which suggested that among the body measurements, shank length best predicted BW with the highest coefficients of determination R2 (adj) in males and females. Abiyu [20] also reported that the best predictor for assessing the body weight of female chickens was back length, keel bone length, and shank length. This could be due to the effects of breeds determining body parts dimensions, environments, and management systems employed [21-25].
In general, fitting the multiple linear regression model to the validation sample can effectively predict BW in local chickens in study districts from their linear body measurements in rural areas, where weighing balance which is used to measure linear body parameters not available for all and is also not used practically when available.
- Ethiopian Biodiversity Institute (EBI). Ethiopian National Strategy and Plan of Action for Conservation and Utilization of Animal Genetic Resources. Addis Ababa: Ethiopian Biodiversity Institute; 2016. Available from: https://www.scirp.org/reference/referencespapers?referenceid=3214559
- Food and Agriculture Organization of the United Nations (FAO). The Second Report on the State of the World’s Animal Genetic Resources for Food and Agriculture. Scherf BD, Pilling D, editors. Rome: FAO Commission on Genetic Resources for Food and Agriculture Assessments; 2015. Available from: https://openknowledge.fao.org/server/api/core/bitstreams/cb3cd3d4-1c97-49e9-b9e6-c6344ad121c4/content
- Getachew B, Kefelegn K, Nagasi A. On-farm phenotypic characterization of indigenous chicken and their production system in Bench Maji zone, southwestern Ethiopia. Afr J Online. 2015. Available from: https://doi.org/10.4314/star.v4i1.10
- Yihun A, Kirmani MA, Molla M. Phenotypic characterization of indigenous chicken ecotypes in Awi zone, Ethiopia. Ecology and Evolutionary Biology. 2020;5(4):131-139. Available from: https://www.sciencepublishinggroup.com/article/10.11648/j.eeb.20200504.13
- Desse T, Melesse A, Taye M. Phenotypic and morphological characterizations of indigenous chicken populations in Kaffa Zone, southwestern Ethiopia. 2018. Available from: https://www.oatext.com/phenotypic-and-morphological-characterizations-of-indigenous-chicken-populations-in-kaffa-zone-south-western-ethiopia.php
- Amine M, Hiskel K, Tekelewold B, Abebe H, Abraham A. Morphometric and morphological characterization of chicken resources adapted to pastoral and agro-pastoral areas of southern Ethiopia. Genetic Resources. 2021;2(4):72-84. Available from: https://orcid.org/0000-0003-3788-4156
- Food and Agriculture Organization of the United Nations (FAO). Phenotypic characterization of animal genetic resources. FAO animal production and health guidelines No. 11. Rome: FAO; 2012. Available from: https://www.fao.org/4/i2686e/i2686e00.pdf
- Dabasa W, Tadesse Y, Shashi B, Singh Y. On-farm phenotypic characterization of indigenous chicken in Dire and Yabello districts, Borena Zone, Oromia Regional State, Ethiopia. J Genet Resour. 2021;7(1):36-48. Available from: https://doi.org/10.22080/jgr.2020.19954.1211
- Wolde B, Tadesse Y, Ameha N. On-farm phenotypic characterization of indigenous chicken ecotypes in West Hararghe zone, Oromia region, Ethiopia. J Vet Med Anim Sci. 2018;2(1):1009.
- Manjura M. Chicken breeding and husbandry practice of the community and morphometric differentiation of local chicken ecotypes in selected district of Gamo Gofa Zone, SNNPR, Ethiopia. Haramaya: Haramaya University; 2019.
- Birhanu A. Production system and phenotypic characterization of indigenous chicken in Awi Zone, Amhara Regional State. Bahir Dar: Bahir Dar University; 2020.
- Nelson NM. Normal eye color in the chicken. Poult Sci. 1947;26:61-66.
- Crawford RD. Origin and history of poultry species. In: Crawford RD, editor. Poultry breeding and genetics. Amsterdam: Elsevier; 1990. p. 1-42. Available from: https://www.scirp.org/reference/referencespapers?referenceid=2192753
- Bekele G, Kebede K, Amaha N. Study of the indigenous chicken production system in Bench Maji Zone, South-Western Ethiopia. Glob J Sci Front Res D Agric Vet. 2016;16. Available from: https://journalofscience.org/index.php/GJSFR/article/view/1748
- Moreda E, Horpal S, Tesefaye S, Anna MJ. Phenotypic characterization of indigenous chicken populations in Southwest and South part of Ethiopia. Br J Poult Sci. 2014;3(1):15-19. Available from: https://idosi.org/bjps/3(1)14/3.pdf
- Tareke M, Assefa B, Abate T, Ermias T. Evaluation of morphometric differences among indigenous chicken populations in Bale Zone, Oromia Regional State, Ethiopia. Poult Sci J. 2018;6(2):181-190. Available from: https://doi.org/10.22069/psj.2018.14974.1329
- Ukwu HO, Okoro VMO, Nosine RJ. Statistical modeling of body weight and linear body measurements in Nigerian indigenous chicken. J Agric Vet Sci. 2014;7:27-31. Available from: https://doi.org/10.9790/2380-07152730
- Singh PN, Mishra AK. Prediction of body weight using conformation traits in Barbari goats. Indian J Small Rumin. 2004;10(2):173.
- Negash F. Predicting body weight of Ethiopian indigenous chicken populations from morphometric measurements. Turk J Agric Food Sci Technol. 2021;9(6):1138-1143. Available from: https://doi.org/10.24925/turjaf.v9i6.1138-1143.4119
- Tadele A, Melesse A, Taye M. Production systems, productive performances, constraints, and rate of inbreeding of indigenous chicken populations in Kaffa Zone, South-Western Ethiopia. J Biol Agric Healthc. 2019;9(5). Available from: https://www.researchgate.net/publication/341713625_Production_Systems_Productive_Performances_Constraints_and_Rate_of_Inbreeding_of_Indigenous_Chicken_Populations_in_Kaffa_Zone_South_Western_Ethiopia
- Moreda E. Phenotypic and genetic characterization of indigenous chicken in Southwest Showa and Gurage zones of Ethiopia. Debre Zeyit: Addis Ababa University; 2015. Available from: https://etd.aau.edu.et/server/api/core/bitstreams/12d9284c-2131-476b-a934-0390e674d236/content
- Mearg F. Production objectives, breeding practices, and selection criteria of indigenous chicken in the central zone of Tigray, Northern Ethiopia. Res J Agric Sci. 2017;5(7):521-528. Available from: https://doi.org/10.14662/ARJASR2017.083
- Fotsa J, Sorensen P, Pym RA. Breeding and reproduction. In: Decision tools for family poultry development. FAO Animal Production and Health Guidelines No. 16. Rome: FAO; 2014. p. 18-25.
- Zereu G, Lijalem T. Production and reproduction performance of local chicken breeds and their marketing practices in Wolaita zone, Southern Ethiopia. Afr J Agric Res. 2016;11(17):1531-1537. Available from: https://doi.org/10.5897/AJAR2015.10728
- Fekede G, Tadesse Y. Breeding practices and preferred traits of indigenous chicken in Western Oromia, Ethiopia. J Livest Sci. 2021. Available from: https://doi.org/10.33259/JLIVESTSCI.2021.85-94
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.



 Save to Mendeley
Save to Mendeley
